PMTA
STATUS
Bidi Vapor submitted its PMTA for all 11 flavors of the BIDI® Stick. The application also indicated its nicotine concentrations of 6% weight/volume as part of its e-liquid formulation.
Last May 2021, Bidi Vapor made it to the FDA’s list of deemed new tobacco products, a list of manufacturers that submitted timely applications under the agency’s PMTA process.
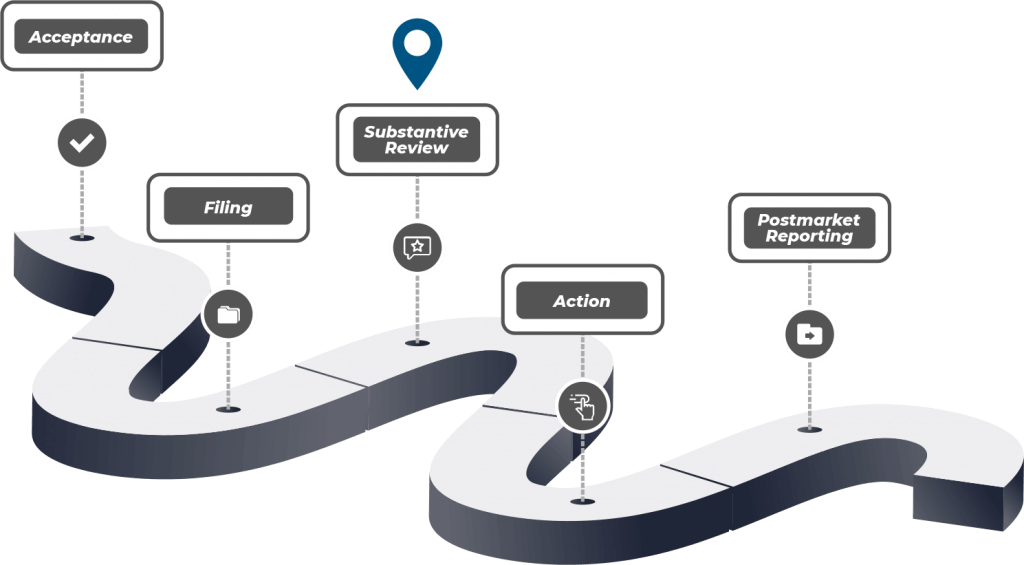
THE PMTA AND WHAT IT MEANS FOR THE VAPE INDUSTRY
Back in 2016, the U.S Food and Drug Administration (FDA) imposed a new rule stating that all Electronic Nicotine Delivery Systems (ENDS) products must be regulated similar to traditional combustible cigarettes.
the PMTA or view more resources,
visit the FDA website now.






How Bidi Vapor Takes Compliance Seriously
Bidi Vapor, the manufacturer of the BIDI® Stick, stands firm with FDA regulations and has always made sure that its products meet quality standards.
In fact, Bidi Vapor’s advocacy in preventing vape products from youth access is evident. “We believe with the proper age-verification restrictions in place, we can meet the needs of the adult smoker while deterring underage use,” states Niraj Patel, Founder and CEO of Bidi Vapor.
From its design and responsible marketing, Bidi Vapor believes it can continue providing adult smokers with a premium vaping experience.


To ensure that only adults 21+ can gain access to Bidi Vapor’s products, we have taken on the challenge and the responsibility to protect the youth against vaping by implementing preventive measures and initiatives.
BIDI VAPOR’S EFFORTS TO RESTRICT UNDERAGE VAPE USE
Website age-gate
To restrict youth from access and exposure to our products, visitors must first input their date of birth before gaining access to the website.
No direct-to-consumer sales
Adult-centric packaging and flavor names
At the company’s expense, the BIDI® Stick was rebranded and repackaged to make it more professional looking and unattractive to underage people. Each flavor name has also been changed to make them sound more generic.
Properly labeled products and marketing materials
Each BIDI® Stick is appropriately labeled to warn against underage use, inform the user of its contents, and instruct consumers on the proper use of the product. All BIDI® Stick marketing materials are labeled with nicotine and underage sale warnings.

